 For all other questions, contact CDC-INFO. Update 1: Your BinaxNOW Self Test May Have a Longer Shelf Life When you reach for your stash of BinaxNOW Self Tests whether youre experiencing symptoms or had a recent exposure its important to ensure your test and the components arent expired.For media inquiries, contact the CDC Newsroom.If you are a clinical or public health laboratory professional, contact LOCS. The BinaxNOW COVID-19 Ag Card Home Test is intended for non-prescription self-use and/or, as applicable for an adult lay user testing another person aged 2 years or older in a non-laboratory setting.Opt in to receive updates from the CDC Laboratory Outreach Communication System. We encourage you to share this message widely with your network(s). Review the bench aids for blood smear preparation, staining, diagnosis, and calculating the percent parasitemiaįor more information, please review the HAN in detail.Ensure that qualified personnel who can perform and interpret these tests are always available.The test targets the histidine-rich protein II (HRPII) antigen specific to Plasmodium falciparum (P. It’s accepted by US and international airlines and cruise lines, with partnerships with United Airlines, American Airlines, and Delta Air Lines. The BinaxNOW COVID-19 Antigen Self Test is intended for non-prescription. As of now, eMed’s BinaxNOW COVID-19 Ag Card Home Test is still the only at-home test approved for returning international flights. Have malaria diagnostic tests available (blood smear or BinaxNow™ rapid diagnostic test followed by blood smear) The BinaxNOW Malaria test is an in vitro immunochromatographic assay for the qualitative detection of Plasmodium antigens circulating in human venous and capillary EDTA whole blood of individuals with signs and symptoms of malarial infection. In Vitro Diagnostics (LIVD) Test Code Mapping for SARS-CoV-2 Tests provided by CDC. Abbott rapid COVID-19 tests are reliable, affordable, and widely available The BinaxNOW COVID-19 Ag Card and the BinaxNOW COVID-19 Ag Card Home Test have received Emergency Use Authorization by the U.S.vivax) in Florida and Texas within the last 2 months, and the concern for a potential rise in imported malaria cases associated with increased international travel in summer 2023.ĬDC recommends that clinical laboratories BinaxNOW COVID-19 Antigen Self Test, 1 Pack, 2 Tests Total, COVID Test With 15-Minute Results Without Sending to a Lab, Easy to Use at Home 12,236 10K+ bought in past month 1988 (9.94/Count) List: 23. It was designed to pair with the NAVICA mobile app, which works like a. Indeed, Prince-Guerra and colleagues noted that while the FDA authorized a number of tests for use in symptomatic persons via emergency use authorization, "data are lacking on test performance in asymptomatic persons to inform expanded screening testing to rapidly identify and isolate infected persons.On June 26, 2023, CDC issued a Health Alert Network (HAN) message about the identification of locally acquired malaria cases ( P. BinaxNOW COVID-19 Ag Card is a digitally-connected rapid test for COVID-19 infection. 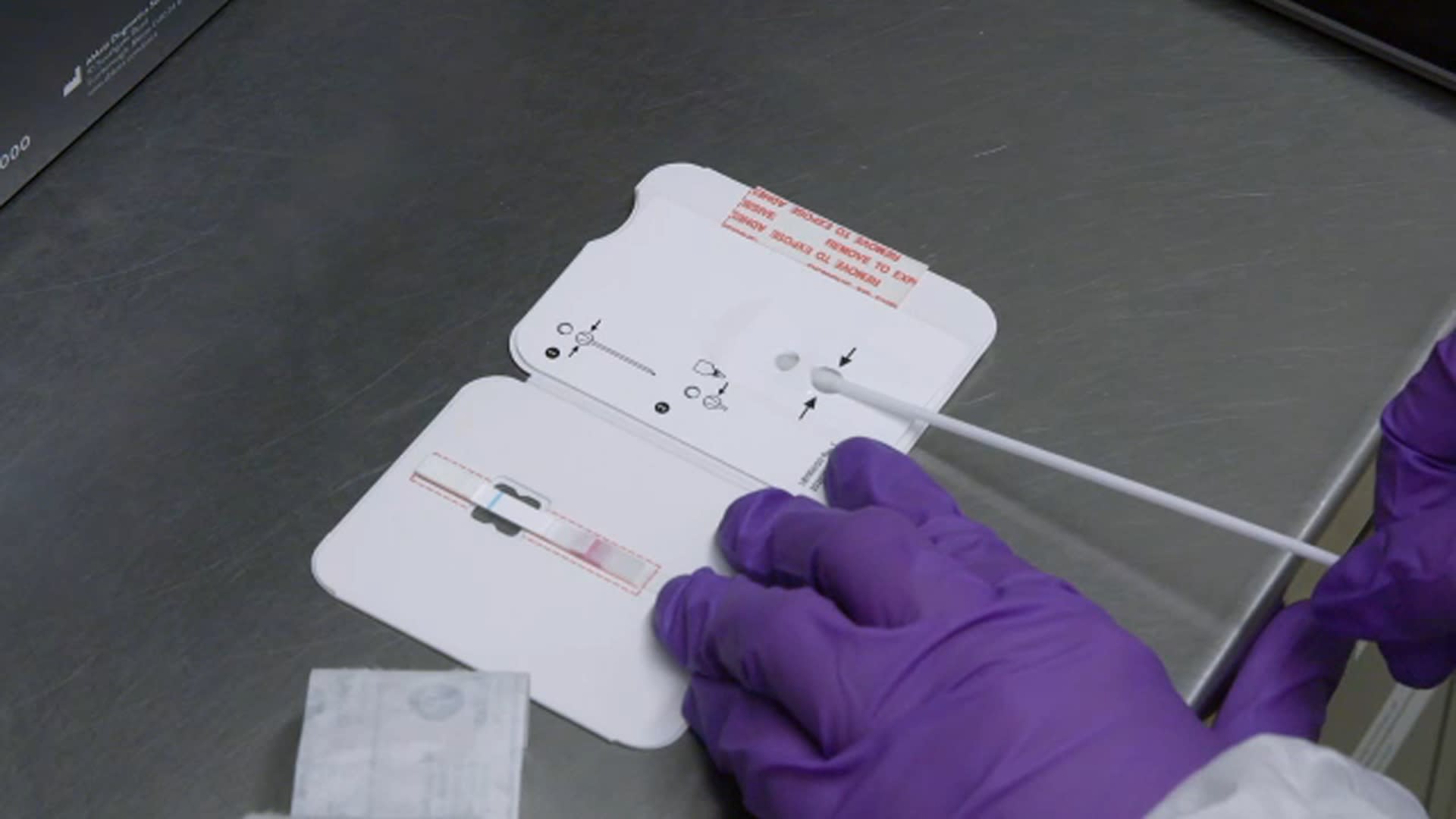
Numerous individuals, including President Trump and first lady Melania Trump, subsequently developed COVID-19 all had tested negative with BinaxNOW as a condition for attending the event (except possibly the president himself, who later acknowledged he seldom underwent testing). Each kit box contains 2 test cards, 2 nasal swabs and 2 reagent bottles. This rapid-result test is for personal use. 
26 Rose Garden event introducing Amy Coney Barrett, who had just been nominated to the Supreme Court. BinaxNOW COVID-19 Antigen Self Test from Abbott can be used to detect active infection, with or without symptoms. Antigen tests are designed to detect proteins from the virus that causes COVID-19 in anterior nasal (nares) swab. According to media reports, a senior Trump administration official said the White House switched to the BinaxNOW test prior to the notorious Sept. The BinaxNOW COVID-19 Ag Card is a type of test called an antigen test. However, they noted that in clinical context, real-time RT-PCR provides "the most sensitive assay to detect infection," and viral culture "is still an artificial system and is subject to limitations."ĭata on rapid antigen testing performance has been sparse, though it has been in the spotlight before. A rapid test for the qualitative detection of COVID-19 antigens in nasal swab specimens Test kit dimensions: 9.125 L x 0.938 D x 5. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
